In a recent interview with Healthcare Brew, Laura Bray discussed how the Iran war could create new vulnerabilities across the global pharmaceutical supply chain, adding pressure to a system already struggling with ongoing drug shortages.
Angels for Change Joins USP Convention
At Angels for Change, we believe that ending drug shortages requires more than urgency, it requires alignment, collaboration, and bold action across every part of the healthcare ecosystem.
That’s why we’re extremely proud to be recognized by the U.S. Pharmacopeia Convention as a voting member among a diverse network of more than 450 organizations working together to strengthen trust in medicines and improve global health outcomes.
Laura Bray presents the 2025 Drug Shortage Guardian Awards at USP.
The USP Convention brings together leaders from across healthcare, science, patient advocacy, academia, government, and industry—each contributing expertise to shape the standards that help improve health through public standards worldwide.
This growing and evolving membership reflects a shared understanding: resilient supply chains and high-quality medicines don’t happen in isolation. They are built through collective responsibility, shared data, and coordinated action.
For Angels for Change, this mission aligns deeply with our own work.
Every day, patients across the United States face treatment delays, substitutions, or interruptions due to preventable drug shortages. Through initiatives like Project PROTECT and our national Drug Shortage Hotline, we’ve seen firsthand that solutions exist when stakeholders come together early, transparently, and with patient impact at the center.
USP’s Convention model reinforces this same principle—bringing together over 450 organizations to guide standards, strengthen systems, and elevate the science behind medicine quality.
Laura Bray and Vimala Raghavendran, Angels for Change's 2025 Change Maker of the Year.
As new members join this community, the opportunity grows stronger: to connect standards with real-world access challenges, to bridge policy with patient experience, and to ensure that quality and availability move forward together—not separately.
At Angels for Change, we are encouraged by this continued expansion of collaborative leadership. Ending drug shortages will require all of us to move in alignment toward a shared goal of reliable access to life-saving medicines for every patient, every time.
We look forward to continued partnership across the USP Convention community and beyond as we work to end drug shortages once and for all.
““The addition of these Members reflects stronger engagement across regions and sectors in advancing quality, supporting a resilient medicines supply chain, and protecting people’s health.””
Convention Member additions
The following organizations have joined the USP Convention as of Spring 2026:
African Medicines Agency (AMA)
Agence Béninoise du Médicament et des autres Produits de Santé (ABRP)
Algeria National Agency for Pharmaceutical Products (ANPP)
Angels for Change
Canadian Generic Pharmaceutical Association (CGPA)
Duke-Margolis Center for Health Policy
Egyptian Drug Authority (EDA)
Indonesian Food and Drug Authority (BPOM)
Institute of Drug Quality Control – Ho Chi Minh City
International Pharmaceutical Students’ Federation (IPSF)
Medicines Control Authority of Zimbabwe (MCAZ)
Pharmaceutical Manufacturers Group of the Manufacturers Association of Nigeria (PMGMAN)
Rwanda Food and Drugs Authority (Rwanda FDA)
Union des Opérateurs Pharmaceutiques (UNOP)
About the USP Convention
The USP Convention is one of USP’s governing bodies, providing input and insights that strengthen USP’s work and impact around the world. Convention membership represents a broad cross-section of the global health ecosystem and includes 450+ member organizations from more than 50 countries, representing expert perspectives of science, manufacturing, regulatory, patient advocacy, government, and academia.
Through the USP Convention, Members elect the Board of Trustees and Council of Experts – USP's other two governing bodies – supporting USP’s mission to protect and promote health by advancing quality across the lifecycle of medicines worldwide.
For additional information, please contact: mediarelations@usp.org
About USP
USP is a private, non‑profit scientific organization that collaborates with stakeholders around the world to develop public quality standards and solutions that support quality across the development, manufacturing, and delivery of regulated health products; strengthen regulatory systems; enhance supply reliability; and support the adoption of innovation. Used in more than 150 countries, USP standards and solutions play a critical role in increasing the availability of quality medicines, dietary supplements, and food ingredients worldwide.
Spotlight on America: Cancer drug shortages leave families waiting; Texas startup tries to fill the gap
This Spotlight on America report follows families like Bryton’s, whose cancer treatment was overshadowed by the fear of drug shortages—forcing them to wait and wonder if life-saving medications would be available. As shortages of essential chemotherapy drugs disrupt care nationwide, the story highlights both the human toll of an unstable supply chain and emerging solutions.
Advocacy Alert: Angels for Change joins Industry Leaders in Official Comment Letters to CMS
Drug shortages affect real patients every single day—making reliable access to medication not just a goal, but a necessity. That’s why we’re proud to champion the patient voice in three important CMS comment letters focused on strengthening a more dependable and resilient drug supply.
At Angels for Change, advocacy is more than a mission—it’s action. By collaborating with leading partners such as the Duke-Margolis Institute for Health Policy, the End Drug Shortages Alliance (EDSA), and the U.S. Pharmacopeia Drug Shortage Task Force, we’re helping drive solutions that are both practical and firmly rooted in the real needs of patients.
Change Maker Chronicle: The March Forth Edition
Project PROTECT Milestone: U.S. Supply of Sterile Water for Injection Protected
Angels for Change is proud to announce a major Project PROTECT milestone: our partner Fagron Sterile Services is now fully production-ready with new U.S. capacity for Sterile Water for Injection—a critical component used in countless injectable medicines.
This marks the second successful Project PROTECT collaboration with Fagron, following earlier investments that helped protect patients during the IV fluid shortage in 2024. By investing in domestic manufacturing infrastructure, we’re strengthening the resilience of the U.S. drug supply and protecting patients before shortages occur.
We don’t just talk about ending drug shortages—we’re creating solutions that build onshore readiness, strengthen supply chains, and protect patient access to lifesaving medicines.
Laura Bray Featured in APIIC White Paper on Strengthening the Drug Supply Chain
Laura Bray and Angels for Change highlighted in the newly released white paper from the API Innovation Center, “Fragility to Resilience: Aligning Investment and Purchasing to Secure America’s Drug Supply Chain.”
Bray emphasized a clear call to action for leaders across the healthcare ecosystem to work together to solve the drug shortage crisis:
“This is not the patient’s job to solve at all… This is a supply chain failure.”
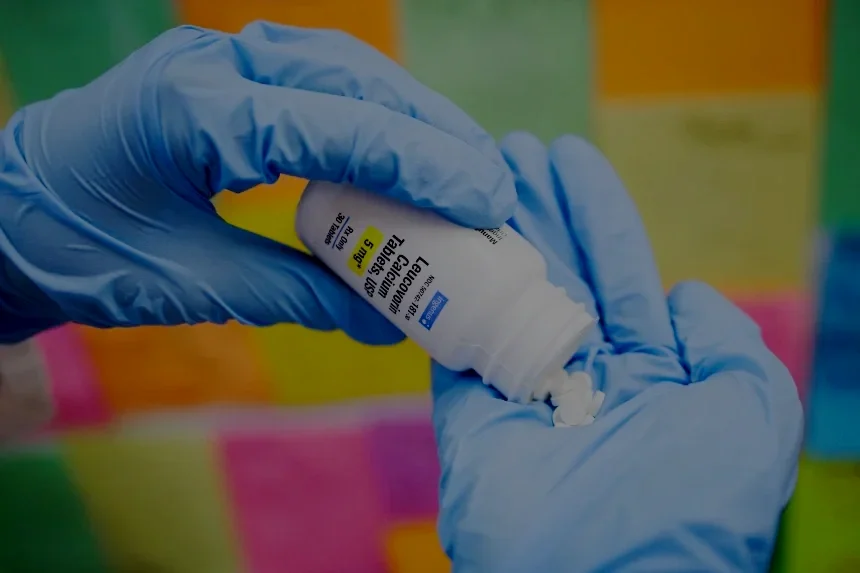
Angels for Change and Project GOLD highlighted in CNN article about the current Leucovorin Drug Shortage in the U.S.
Drug shortages are making national headlines again as families search for critical medicines like leucovorin. In a recent CNN article, Angels for Change founder Laura Bray highlights how the crisis was predictable—and preventable. Through Project GOLD, Angels for Change works across the supply chain to locate scarce medications and help patients access the treatments they depend on, because no family should have to face a shortage alone.
Angels for Change serves as Advising Member in the Drug Resiliency & Transparency Pilot
At Angels for Change, we are proud to participate in the important work of the End Drug Shortages Alliance — serving as both an EDSA member organization and advising partner in the Drug Resiliency & Transparency Pilot.
The initial phase of the pilot demonstrated a powerful truth: when leaders come together early through trusted, neutral governance, transparency delivers real planning value.
Project PROTECT Milestone: Angels for Change and Mark Cuban Cost Plus Drug Company have Strengthened the U.S. Supply of Carboplatin & Methotrexate
Angels for Change today announced that the Mark Cuban Cost Plus Drug Company has completed the Preparation and Protective phases of Project PROTECT for Carboplatin Injection and Methotrexate Injection—two essential oncology medicines currently in nationwide shortage. This milestone strengthens onshore readiness and improves supply availability for patients across the United States.